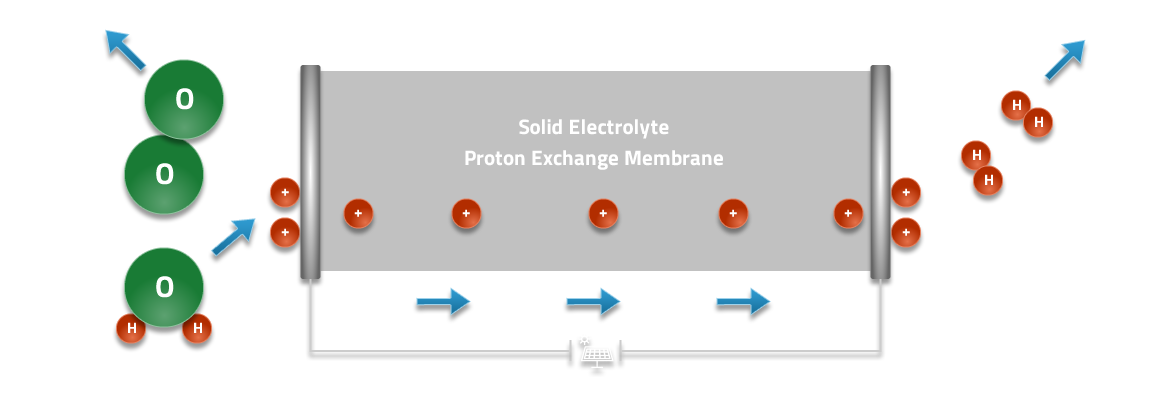
Electrolysis
In electrolysis mode water enters the cell and is split at the surface of the membrane to form protons, electrons, and gaseous oxygen. The gaseous oxygen leaves the cell while the protons move through the membrane under the influence of the applied electric field while electrons move through the external circuit. The protons and electrons combine at the opposite surface to form gaseous hydrogen. The hydrogen and oxygen can be produced at pressure in the electrolysis cell, minimizing the need for external mechanical compression.
PEM Electrolysis Process

A PEM electrolyzer consumes power and water to produce hydrogen, oxygen, and heat.














